TECHNOLOGY
Monoclonal antibodies (mAbs) have emerged as a growing class of oncology therapeutics.
Despite their success in many areas, the therapeutic efficacy of mAbs is limited, with only a minority of patients responding to these agents as monotherapies. It is becoming clear that many tumors evade successful elimination by co-opting the body’s own immune regulatory systems.
Our lead protein is a molecule that can improve the therapeutic effects of mAbs by:
- Increasing the sensitivity of cancers to treatment
- Unmasking resistant cancers so they again become sensitized
- Potentially allowing treatment of cancers that have become refractory to therapy.
Our lead product, Ad35K++, is a synthetic protein derived from the CD46-binding sequences of rare human adenoviruses. We have genetically engineered Ad35K++ so that it would bind to CD46 with 25-fold higher affinity than the native protein giving it an extremely high affinity as shown by a picomolar dissociation constant (Kd). The molecule crosslinks several CD46 proteins on the membrane of cancer cells, and leads to internalization of CD46 and its subsequent degradation inside the cell. Ad35K++ binding results in transient removal of CD46 from the surface of lymphoma cells (and a series of other tumor cells, including breast and colon cancer cells) for about 72 hours after treatment. It is an attractive therapeutic product for commercialization based on its ease of manufacturing, low cost of goods, and potential combination with multiple therapeutic mAbs. To our knowledge, no other therapy is in development that downregulates CD46 in order to sensitize tumors to the action of mAbs.
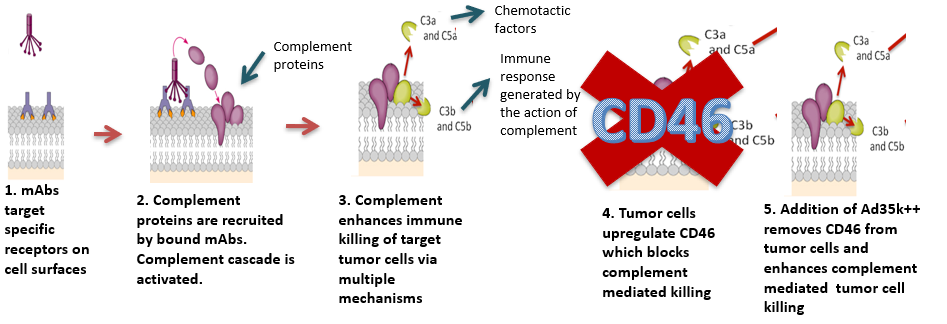
Our first candidate, CMP-001, is a combination of our Ad35K++ technology with an anti-CD-20 antibody. The 1-, 5-, and 10-year survival rates for B cell non-Hodgkins lymphoma (NHL) are 79%, 63%, and 51%, respectively. The anti-CD20 antibody rituximab has become a cornerstone of nearly all therapies for NHL. In most cases of B cell NHL recurrence, patients became resistant to rituximab therapy. Although the 5-year survival rate for B cell NHL has increased from 48% (1975-1977) to 64% (1996-2003) since the introduction of rituximab, there is still substantial room for improvement. One of the mechanisms of action of rituximab involves binding to lymphoma cells and subsequent activation of the complement system, a cascade of proteins in the blood that normally participates in the elimination of pathogens from the host. Complement activation then leads to the killing of lymphoma cells. One of the mechanisms by which lymphoma cells actively block this activity of rituximab is by up-regulating CD46 and preventing killing by the complement system. Increased levels of CD46 are found on the membranes of lymphoma cells and may contribute to the ineffectiveness of rituximab in treating NHL.
We have shown that CD46 expression on leukemia cells was more uniform and at least one order of magnitude higher than on normal PBMCs. These findings are in agreement with our previous study with samples from 38 multiple myeloma patients in which we documented that the mean CD46 receptor numbers on neoplastic plasma cells and normal hematopoietic cells were 49,130/cell and 7,340/cell, respectively. This trend is also seen in other cancers.

Our studies indicate that many mAbs can kill tumor cells in vitro by using complement. To date, we have shown that Ad35K++ significantly enhances complement dependent killing triggered by rituximab (Rituxan®), alemtuzumab (Campath®), trastuzumab (Herceptin®), and cetuximab (Erbitux®) and others as shown below. No Ad35K++ associated cytotoxicity was observed on tumor cells lines that did not express the target molecule for the corresponding mAb.

